We observed that the prion protein progressively converts to the pathological form and induces neuronal loss in the brain. To shed light on how the prion protein changes from its normal to the disease-causing form, we expressed the prion protein from hamster in transgenic flies. An abnormal form of the prion protein is not only responsible for the disease in several mammals, but is also an infectious agent that can transmit the disease within or across species. This disorder belongs to a unique class of degenerative diseases that includes mad-cow disease in bovine and scrapie in sheep. 
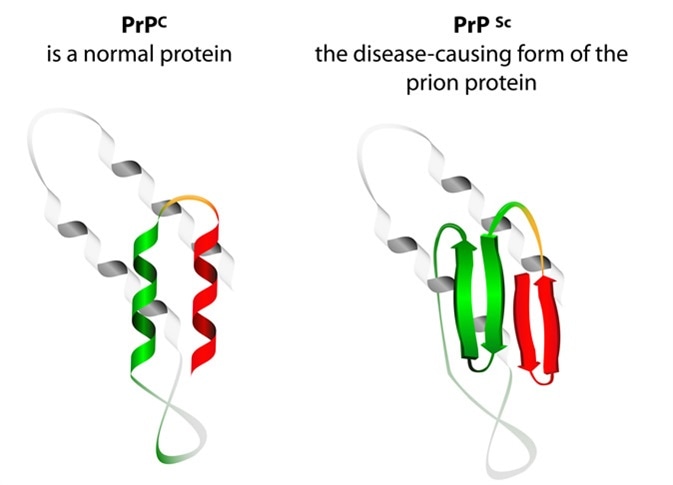
Overall, we provide new insight into the mechanisms of spontaneous accumulation of neurotoxic PrP and uncover the potential therapeutic role of Hsp70 in treating these devastating disorders.Ĭreutzfeldt-Jakob disease is a type of dementia caused by the deposition of the prion protein in the brain. Moreover, we identify a new role for Hsp70 in the accumulation of misfolded PrP. These results highlight the ability of wild-type PrP to spontaneously convert in vivo into a protease-sensitive isoform that is neurotoxic, supporting the idea that protease-resistant PrP Sc is not required for pathology. This protective activity involves the direct interaction between Hsp70 and PrP, which may occur in active membrane microdomains such as lipid rafts, where we detected Hsp70. Remarkably, Hsp70 prevents the accumulation of PrP Sc-like conformers and protects against PrP-dependent neurodegeneration. Next, we investigated the role of molecular chaperones in PrP misfolding in vivo. Thus, wild-type PrP rapidly converts in vivo into a neurotoxic, protease-sensitive isoform distinct from prototypical PrP Sc. In contrast to PrP Sc from mammals, PrP is proteinase-sensitive in flies. Aged flies accumulate insoluble PrP that resists high concentrations of denaturing agents and contains PrP Sc-specific conformational epitopes. In young flies, PrP demonstrates properties of the benign PrP C in older flies, PrP misfolds, acquires biochemical and structural properties of PrP Sc, and induces spongiform degeneration of brain neurons. To shed light on how wild-type PrP undergoes conformational changes and which are the cellular components involved in this process, we analyzed the dynamics of wild-type PrP from hamster in transgenic flies. In humans, prion disorders, such as Creutzfeldt-Jakob disease, present typically with a sporadic origin, where unknown mechanisms lead to the spontaneous misfolding and deposition of wild type PrP. Prion diseases are incurable neurodegenerative disorders in which the normal cellular prion protein (PrP C) converts into a misfolded isoform (PrP Sc) with unique biochemical and structural properties that correlate with disease.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
